Nanoscale chemical imaging reveals the mechanism of action of high-voltage additives on the positive electrode surface interface
Prof. Yang's group has recently collaborated with Prof. Zhou Jigang, Energy Storage Group, Canadian Light Industry Applications Division, in the study of nanoscale Chemical imaging, revealing the mechanism of high voltage additives on the surface of positive electrode. The results are published in Chemical Communications, 2017, 53,8581。
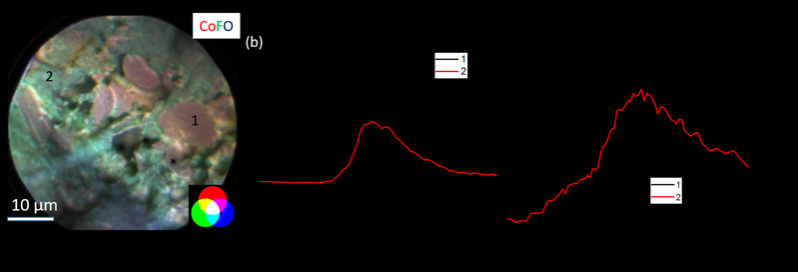
Improving the energy density of lithium-ion batteries is a key step for their eventual large-scale application in energy storage and transportation. The application of high voltage positive electrode can greatly improve the energy density of the battery but has little effect on the process route, so it has attracted extensive attention in the battery field. Improving the energy density of lithium-ion batteries is a key step for their eventual large-scale application in energy storage and transportation. The application of high voltage positive electrode can greatly improve the energy density of the battery but has little effect on the process route, so it has attracted extensive attention in the battery field. In close cooperation with Dr. Jigang Zhou of the Canadian Light Source Energy Storage Group and Dr. Jian Wang of the Chemical Imaging Line Station, the photoelectron microscopy (X-PEEM) technique, which is traditionally used to study smooth surfaces, was creatively used to study the chemical and electronic structure characterization of the surface of high-voltage lithium cobaltate cathode by additives. X-PEEM uses advanced synchrotron radiation sources for chemical imaging at the nanoscale, with elemental and orbital selectivity, chemical and electronic structure sensitivity, and surface sensitivity. This study provides the first practical imaging of the surface chemistry and electronic structure of a composite porous electrode at the 100nm scale. The effects of the additives on various components of the electrode, including lithium cobaltate, conductive carbon black and the surface of the binder, and their interrelationships are revealed. In this study, it was found for the first time that the additive had an effect not only on the composition and content of the film on the electrode surface, but also on the distribution of COF2, an important film forming component. The use of additives allows this protective component to be deposited preferently on the surfaces of binders and conductors, thus protecting the stability of these important electrode components at high voltages. The study also found that the use of the additive reduced the covalent bond between the cobalt and oxygen and thus better stabilized the electrode surface. These findings have important guiding significance for the further optimization of additives and electrolytes. This work has also opened the way for nanochemical imaging to study complex electrode systems without the need for traditional sample processing in order to maximize the preservation of the original information of the electrode (equivalent to near-in-situ studies of practical composite electrode surface structure imaging). This method is suitable to be extended to other electrode systems for the study of reaction mechanism and attenuation mechanism.
Co-first authors of the paper are Dr. Yajuan Ji of Xiamen University and Dr. Jian Wang of the Canadian Light Source Energy Storage Group. The Laboratory of Physical Chemistry of Solid Surfaces of Xiamen University is the common communication unit. This work was supported in part by the Key Project of National Natural Science Foundation of China (No. 21233004) and NKRDP (No.2016YFB0901502).
Paper link:http://pubs.rsc.org/en/content/articlepdf/2017/CC/C7CC03960C
