Important progress has been made in the synthesis of high nickel cathode materials for lithium-ion batteries
Prof. Yong Yang's research group has recently worked closely with Dr. Feng Wang of Brookhaven National Laboratory and Dr. Khalil Amine of Argonne National Laboratory to make important progress in the synthesis of high-nickel cathode materials for lithium-ion batteries. The results have been published as a paper on Advanced Material, 2017, 1606715.
The development of a battery system with an energy density of up to 300Wh/kg is a new generation of lithium ion batteries that attracts much attention in the world, and the high nickel cathode material is an essential cathode material system. However, due to the difficulty in synthesis of high nickel cathode materials (especially when the nickel content is higher than 0.7), the capacity utilization and cycle stability of high nickel cathode materials are prone to poor performance. The research work through a variety of spectroscopy characterization techniques, such as in situ X-ray diffraction, X-ray absorption spectrum, neutron diffraction spectrum and high resolution solid state nuclear magnetic spectra confirmed control suitable synthetic temperature (~ 850 oc) and reaction time, and can get good crystalline, ion LiNi0.7 low degree of mixed Co0.15 Mn0.15 O2 cathode material, within the range of 3.0 4.3 V voltage, current density under the condition of 10 ma/g, its reversible discharge capacity is 197 mah/g, the cycle of 50 weeks retention capacity can be as high as 93.5%.
The reversible discharge capacity is as high as 240mAh/g in the potential range of 3.0-4.5V. The effects of various preparation conditions on the grain growth and cation mixing process of high nickel materials are also discussed in detail by proper thermodynamic and kinetic analysis (see the figure).
The first author of this paper is Dr. Dawei Wang, who graduated from Professor Yang Yong's research group in 2015. Xiamen University is the common communication unit. This work was supported in part by the National Natural Science Foundation of China (No. 21233004), the Interfacial Electrochemistry Innovation Group of the National Natural Science Foundation of China (No.21621091), and the National Key Basic Research and Development Program of China (No.2016YFB0901500).
Paper link:onlinelibrary.wiley.com/doi/10.1002/adma.201606715/full
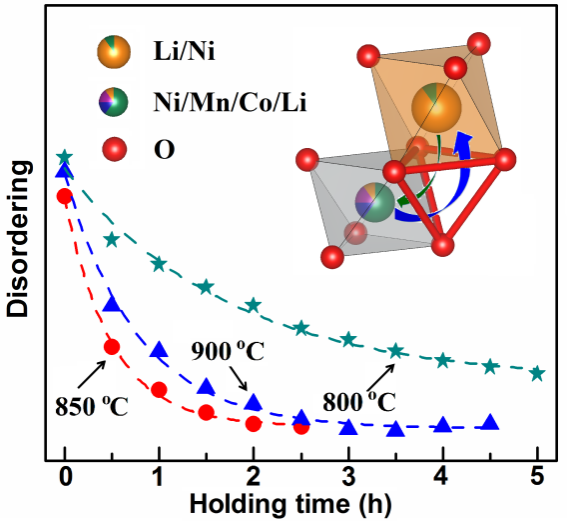
The figure shows the variation characteristics of the disorder/cation mixture arrangement of NCM71515 material with reaction time at different temperatures
